Relaxip is fantastic!!! Because...
Relaxip


About Yoko Katz, a Founder
Breast Cancer Survivor / Recurrence Fighter
Economics Professor at Fashion Institute of Technology
Fashion Economist
and Hat and Clothes Designer
Fashion for Cancer Patients
When Yoko went through breast cancer for the first time in 2014, the images of cancer patients being skinny, and miserable scared her. She learned that cancer patients could keep their jobs while going through chemotherapy. She kept teaching at Fashion Institute of Technology. She wanted to be herself and look normal by seeking help in fashion. When she bought her first wig in orange, she started to document her fashion and created the blog Heal in Heels.
Our Story
① June 2014
First ever surgery: Mastectomy
Age of 36. Nobody choses to have cancer. I had a healthy lifestyle with no familly history of breast cancer but I still found a lump on me. After the mastectomy I found that I coulden’t raise my arms and that was painful and frustrating to me. I coulden’t even wear a T-shirt but I still had to wear something to go to the doctors office.
② June 2014
The Birth of Original
Going out with my surgical tubes on me. During my post-surgery, checking my blood for egg retrieval, and meeting with my reconstruction surgeon, I cried in pain because I could not wear a T-shirt. My mom came from Japan to assiste me during treatment, she came with a special shirt and told me to try it on. I could wear it without pain and without raising arms. My surgeon saw me putting it on, saying “Wow, Yoko, I have never seen anything like that.” The birth of the first Relaxip.
③ Fall 2018
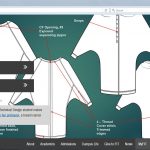
My student, Sidney, asked if I had some issues in garments during recovery.
End of Fall 2018 Semester. While teaching at the Fashion Institute of Technology, I did presentations about my cancer story. Asking fashion students to be creative in helping society. Sidney asked if I had some issues in garments during recovery. We decided to work together for a thesis project for her Technical Design degree.
④ Spring 2018
Meeting with Sidney, almost every week
We developed 3 garments for breast cancer patients. Sidney was so enthusiastic about making this project successful. The course requirement was to make one garment, instead she was willing to work on three. By the end of the semester, her presentation at FIT was well received. We got featured in the FIT website news page and its publication, HUE magazine, winter 2018 issue.
⑤ Summer 2018 - Summer 2019
Moving one of the three designs into production.
Searching for meaning in cancer treatments. There was no answers to why I got breast cancer. Cancer controlled me and my world became upside down. I could not change my environment nor my body, but I could control one thing, I can still choose who I want to be and how I want to do things. I wanted to choose fashion to help cancer patients, which back in 2014, there were none. I wanted to contribute to the world to shift some images of cancer patients’ life. Relaxip means a lot to me and I want to help those who are going through the same thing as me. Starting up a business has been taken a lot of learning and I couldn’t have done it without Masayo Kawanishi, who is my friend.
⑥ September 2019
The very first production is ready.
I was told a recurrence after 5 years.
Ironic Moments. After thinking about this prduct for 5 years, I am finally ready to send them off and help many patients. Then, I was told that my cancer came back. What? Now? It cannot be. My goal for Relaxip is to help other people, but it turns out that I might be the very first patient to use my own product.
Photo Credit: Dennis Cahlo
Some other notes

Estimated New Breast Cancer Diagnoses for 2019
Other Potential Use
Relaxip will serve many other occasions
- Shoulder Problems
- After surgery for lung, heart and other upper body problems
- People who needs to carry surgical drains
- People who needs to have easy access to treatments at hospitals
- Relaxip is an adaptive clothing
SHOP
Our first production garments are on sale!
Due to a preliminary webpage for the fundraising campaign, the shop is in progress and will be structured better in October.
Blue with Gray Zippers
$ 69.50
Available in S/M (dress size 0-6)
Available in L/XL (dress size 8-12)
Navy with Gray Zippers
$ 69.50
Available in S/M (dress size 0-6)
Available in L/XL (dress size 8-12)
Navy with Pink-Purple Zippers
$ 69.50
Available in S/M (dress size 0-6)
Available in L/XL (dress size 8-12)
*We will donate a part of our proceeds to immuno therapy research institution for breast cancer.
Stay in touch with us with SNS
Media Coverage
Book: Tough
SHARE Support
JP Magazine
FIT Article
Muscal: To Live
German Radio
My Heal in Heels information is now moved to “Blog” section in the latest website.